Do you know what the most prevalent component in your skincare is? Surfactants! Surfactants may be found in a wide range of cosmetics and personal care products, from cleansers to moisturizers and makeup.
Stay with us to the end to find out why and whether you should buy products containing surfactants. Let’s get started.
Highlights:
- What Are Surfactants?
- Science Of Surfactants
- How Are Surfactants Made?
- Different Types Of Surfactants
- Common Surfactants Used In Cosmetics
- Role Of Surfactants In Skincare
- Alternative Surfactants For Your Skin And Hair
- Interactions Between Surfactants And The Skin
What Are Surfactants?
Surfactants are a key ingredient in cleaning detergents. Surfactants, as the name implies, elicit action on the surface being cleaned in order to capture debris and remove it from the surface.
Surfactants are found in soaps, shampoos, and hair conditioners because they help cleanse your scalp, skin, and hair. When these chemicals are combined with water, they produce a nice frothy response. They are typically suitable for all skin types.
Surfactants are employed in lubricants, inks, anti-fogging solutions, herbicides, adhesives, emulsifiers, and fabric softeners in addition to soaps and detergents.
Science Of Surfactants
Let’s take a closer look at how surfactants function chemically. Surfactant is an abbreviation for surface active agents. Surfactants work at interfaces such as the oil-water or air-water contact. It changes the surface tension (molecular forces) of the two fluids. How do they accomplish this?
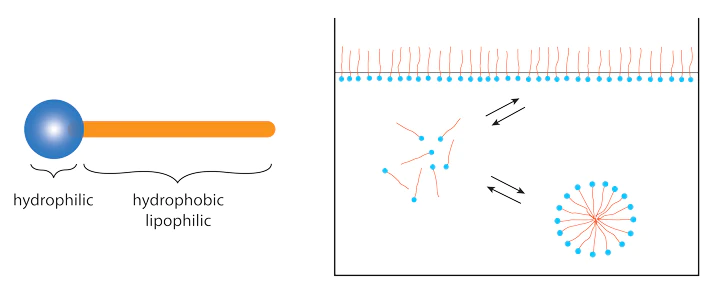
Surfactants are amphiphilic molecules (amphi: both + philia: love), which means they have two ends:
- A hydrophilic head (hydro: water + philia: love) is the water-loving end that aligns with water molecules while moving away from oils and fats.
- A hydrophobic tail (hydro = water + phobia = fear) – this end of the molecule travels away from water and often aligns with oils and fats.
When adequate quantities of surfactants are introduced to a solution, they rearrange in such a way that the hydrophilic heads face the water. The hydrophobic tails attract oil or debris, creating droplets, which are also known as micelles. (as shown in the image below). Surfactants with this chemical characteristic can be good detergents, emulsifiers, or foaming agents.
How Are Surfactants Made?
Petrochemicals are commonly utilized as starting materials (reactants in chemical processes) in the production of synthetic surfactants. They are then subjected to chemical processes such as sulfonation (sulfur addition) or ethoxylation (addition of ethylene oxide). Because they are synthetic in nature, they may be developed or blended with other substances to get the desired results.
Yet, there are a variety of biosurfactants that offer significant benefits as an environmentally benign alternative to synthetic surfactants. These are microorganism-produced compounds with well defined hydrophilic and hydrophobic groups.
Different Types Of Surfactants
Surfactants are classified into four classes based on the charge on their hydrophilic heads.
Nonionic Surfactants
Nonionic surfactants, as the name indicates, have no net charge in solution. Since they are gentle, they are frequently used in cosmetics that lack foaming or lathering qualities.
Nonionic surfactants can be efficiently mixed with other surfactant types. They are widely found in hand and body lotions. Stearyl alcohol and cetearyl alcohol are two typical nonionic surfactants found in skincare products.
Anionic Surfactants
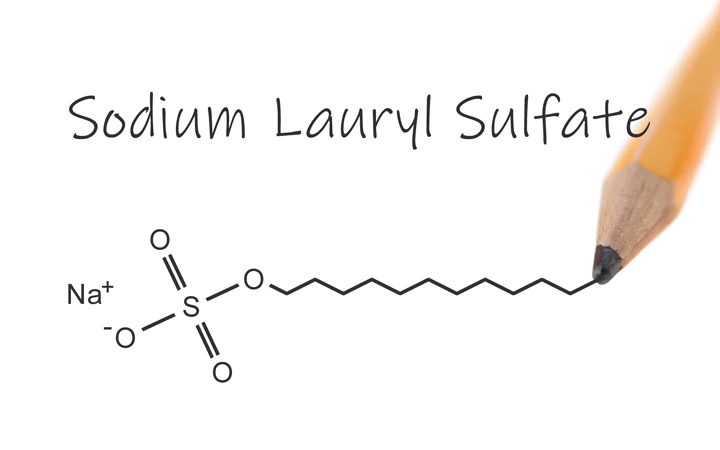
They are negatively charged surfactants that are effective in removing oil and grime from the surface of your skin. Anionic surfactants, which have high cleaning properties, are the most often used kind as major detergents in soaps, shampoos, and cosmetics. They can, however, be harsh and unpleasant to your skin.
To reduce the harshness, such surfactants are frequently mixed with amphoteric or nonionic secondary detergents. SLS (sodium lauryl sulphate), sodium stearate, alpha olefin sulfonate, and other chemicals are examples.
Cationic Surfactants
Since they are positively charged, these surfactants are ineffective as detergents and are hence not employed in cleansers. The majority of human skin is negatively charged. As a result, cationic surfactants have a great affinity for it. Nonetheless, because of this feature, surfactants are good transporters of therapeutic treatments for damaged skin and hair.
Fabric conditioners also include cationic surfactants. Among the most frequent cationic surfactants are quaternary ammonium compounds.
Zwitterionic/Amphoteric Surfactants
The presence of both positive and negative charges in solution gives rise to the surfactant’s name. They have the ability to change the pH of the environment in which they are introduced. These surfactants might be soothing to your skin.
Amphoteric surfactants may be used in skincare products as secondary surfactants to minimize irritation and promote foaming. Amphoteric surfactants include betaines, sulfobetaines, some amino acids, and phospholipids.
Common Surfactants Used In Cosmetics
The following are some common surfactants used in skin and hair care products, along with their functions:
| Surfactant | Function |
| SLS (Sodium lauryl sulphate) ALS (Ammonium lauryl sulphate) Olefin sulfonates | Cleansers, foaming agents |
| Sodium stearate | Soap |
| Stearic acid | Deodorants, antiperspirants |
| Cetrimonium chloride Stearalkonium chloride | Hair conditioners |
| Sodium Lauriminodipropionate Disodium Lauroamphodiacetate | Mild cleansing agents for sensitive skin and hair |
| Cetyl alcohol Stearyl alcohol | Emulsifiers, thickening agents |
| Cocamidopropylamine oxide | Foaming agent |
| Polysorbate ester | Solubilising agent |
Role Of Surfactants In Skincare
Surfactants are one of the most common substances used in the cosmetics industry. Because of their diverse chemical characteristics, they are highly useful in personal care products.
Let’s look at some typical surfactant usage in the cosmetics business.
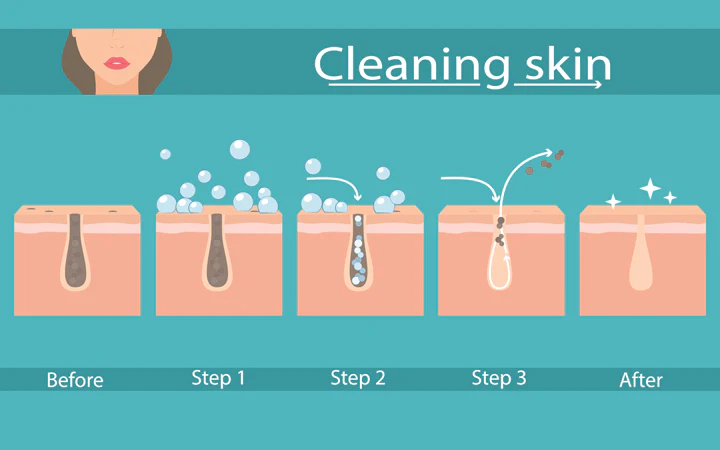
1. Detergents or Cleansers
Surfactants, due to their amphiphilic nature, can produce micelles that trap oil, debris, or sebum. It loosens them from the surface of your skin, allowing them to be easily wiped away. They can be found in soaps, shampoos, and face washes.
2. Emulsifiers
Surfactants can be employed as emulsifiers, which help to keep a mixture of two immiscible or insoluble liquids, such as oil and water, stable for a long time. Emulsifiers are widely found in creams, lotions, conditioners, and other cosmetic products.
3. Foaming Agents
Surfactants in this class lower surface tension at the air-water contact. They also improve the development of lather or bubbles. These surfactants can be found in items such as shaving creams, which assist soften the stubble for a smooth shave.
4. Thickeners
Some surfactants have the ability to interact with other chemicals in a formulation, trapping them in a network of main surfactant molecules. This causes the goods to thicken. They are commonly used in the production of thick winter body lotions, conditioners for dry hair, mascaras, and other cosmetics.
5. Wetting Or Dispersing Agents
The intramolecular tensions at the liquid interface are reduced by these surfactants. They also help products containing them disseminate and penetrate deeper into the skin and hair. Surfactants in cosmetic products are most typically utilized as wetting or dispersion agents.
6. Opacifiers
Certain surfactants can make a formulation opaque by absorbing light and making the surface applied on appear brighter. They are widely utilized in cosmetics recipes.
7. Conditioners
Cationic surfactants can build a tough, protective coating on the surface of the skin or hair. They are often found in cosmetics, hair conditioners, and other similar items.
8. Preservatives
Surfactants can be utilized as preservatives in cosmetics due to their antibacterial characteristics.
Alternative Surfactants For Your Skin And Hair
While selecting surfactants, exercise caution. They are not only a vast range of chemicals, but they are also utilized in conjunction with other surfactants and other compounds. The interplay of all the chemicals produces the surfactant’s ultimate impact on your skin.
Avoid using strong chemicals like SLS or SLES (Sodium laureth ether sulfate). Strong surfactants can deplete your skin’s natural moisture and expedite the aging process.
Another class of surfactants, such as PEG (polyethylene glycol), can function as transporters for possible carcinogens in the skin (cancer-causing).
Natural surfactants made from plants are considered to be safer to use. They are subjected to certain chemical processing in order to be appropriate for usage in cosmetics. Natural surfactants include potassium cocoate (produced from coconut oil), decyl glucoside (derived from maize and coconuts), sucrose cocoate (derived from sugar beets), and others.
The majority of SkinKraft’s personalized skin and hair care products use natural surfactants sourced from coconut or palm oil, as well as maize glucose. They gently cleanse your face and hair without removing vital oils.
Interactions Between Surfactants
Surfactants in a product can primarily interact with the skin in two ways:
1. Interaction with SC
Surfactants can interact with the stratum corneum (SC), the skin’s outermost layer. Surfactants combine to generate monomers or polymers that can penetrate the skin barrier and profoundly hydrate.
Nevertheless, using strong or harsh surfactants for an extended period of time might permanently damage the cell structure of your skin’s surface. This can harm your skin’s barrier function and lead to inflammation.
2. Altering the pH
Surfactants can influence the pH of your skin’s surface depending on their net charge following dissolution. Items with high pH values can cause dry, itchy skin and acne outbreaks.
Employ formulas with a pH that is adequately balanced. Acne outbreaks can be reduced temporarily by using moderately acidic cleansers. Cleansers that are slightly alkaline might assist if you have eczema or dermatitis.
Wrapping Up
Surfactants are absolutely necessary in our daily life. Surfactants help cleaners function better, whether you’re cleaning dishes, scrubbing your skin, or doing laundry. As a result, the safest approach is to be aware of the surfactants included in the items you use on a regular basis. Whenever possible, utilize the milder versions. Likewise, try purchasing products from producers that explicitly state that their goods are devoid of harsh ingredients such as SLS and parabens.
















